Spravato (esketamine) is a prescription drug approved to treat specific forms of depression. It comes as a nasal (nose) spray. Spravato is usually given on a dosing schedule that varies by the condition it’s prescribed to treat.
Spravato is prescribed for treating the following conditions in adults:
The active ingredient in Spravato is esketamine. (The active ingredient is what makes the drug work.)
Spravato is a kind of antidepressant. Specifically, it belongs to a group of drugs called NMDA receptor blockers.
This article describes the dosages of Spravato, as well as its strength and how to take it. To learn more about Spravato, see this in-depth article.
Below is some general information about the standard dosage of Spravato.
What is Spravato’s form?
Spravato comes as a liquid solution in a nasal spray device. You take it by spraying it into your nose.
What strength does Spravato come in?
Spravato comes in one strength of 28 milligrams (mg).
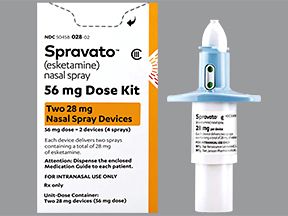
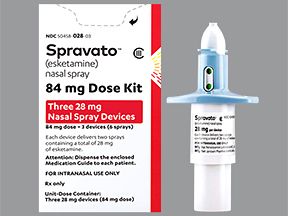
Spravato is available in a 56-mg dose pack that includes two 28-mg nasal sprays. It’s also available in an 84-mg dose pack that includes three 28-mg nasal sprays.
What are the usual dosages of Spravato?
The information below describes the strength and dosing frequency most commonly prescribed or recommended. Your doctor will determine the best dosage to fit your needs.
Each nasal spray device has 28 mg of Spravato and contains two sprays. You will use more than one device to get your full dose. For example, you’d use two devices (four sprays) to get a 56-mg dose of the drug.
Dosing schedule for treatment-resistant depression
Treatment-resistant depression (TRD) is depression that doesn’t improve even after you’ve tried at least two different antidepressants. If you take Spravato for this condition, your dosing schedule will have an induction phase and a maintenance phase.
When you start Spravato, you’ll follow a schedule called the induction phase. This dosing schedule lasts 4 weeks. It helps your doctor safely figure out what the best dose is for you.
After the induction phase, you and your doctor will decide whether to continue Spravato treatment. It depends on how well Spravato has helped your TRD and how well you tolerate the medication.
If you continue treatment, you’ll follow a schedule called the maintenance phase. In this phase, you’ll take doses less often than in the induction phase. Your doctor will prescribe the maintenance dosage of Spravato that’s best for you.
Typical dosages for induction (starting) phases and maintenance (ongoing) phases are shown below.
| Phase | Dose | Frequency |
| Induction | ||
| Weeks 1*–4 | 56 mg or 84 mg | twice per week |
| Maintenance | ||
| Weeks 5–8 | 56 mg or 84 mg | once per week |
| Weeks 9 and on | 56 mg or 84 mg | once every 1 or 2 weeks |
* The first dose of Week 1 is 56 mg.
You’ll usually take Spravato along with another antidepressant, which typically comes in the form of a pill that you swallow.
Dosing schedule for major depressive disorder with suicidal thoughts and behavior
Major depressive disorder is sometimes referred to as depression. One kind of depression includes a sudden onset of suicidal thoughts or behaviors.
The usual dosage of Spravato for this condition is 84 mg twice per week for 4 weeks.
If you have any bothersome side effects, your doctor may lower your dosage to 56 mg twice per week.
Spravato is not usually prescribed as a long-term treatment for this condition. Your doctor will determine whether you will keep taking Spravato after 4 weeks.
You’ll usually take Spravato along with another antidepressant, which typically comes in the form of a pill you swallow.
Is Spravato prescribed long term?
You may or may not take Spravato long term. This will depend on the condition you are taking Spravato to treat and how your body responds to the drug. Talk with your doctor about whether Spravato is a good long-term treatment option for you.
Dosage adjustments
Spravato dosing is not adjusted based on liver or kidney problems. But if you have liver problems, your doctor might monitor you for a longer period of time after each dose of Spravato.
Doctors usually adjust Spravato based on how well it works and whether it causes side effects.
Below are answers to a few commonly asked questions about Spravato.
Is the dosing schedule for esketamine the same as for Spravato?
Yes, there is only one dosing schedule right now. Esketamine is the name of the active ingredient in Spravato. There isn’t a generic version of esketamine, so the only dosing schedule is for the brand-name drug, Spravato.
What is the Spravato REMS program?
The risk evaluation and mitigation strategy (REMS) program is a safety program run by the Food and Drug Administration (FDA). You and your doctor will have to agree to follow certain rules in order for you to safely take Spravato.
The following are examples of rules in the REMS program:
- You’ll stay at the healthcare facility for at least 2 hours after your dose.
- You’ll have someone give you a ride home.
- You won’t drive or do other tasks that require an alert mind on the day of your dose.
Learn more about the Spravato REMS program on the program’s website or call 855-382-6022. And talk with your doctor if you have questions about the REMS requirements.
Is Spravato the same thing as ketamine?
No, Spravato is not the same drug as ketamine. The active ingredient in Spravato is esketamine. There is some esketamine found in ketamine, but they are two separate drugs prescribed for different purposes. For example, ketamine is prescribed as an anesthetic for surgery, and esketamine is not.
Learn more about the differences in the “FAQ” section of this article.
You’ll take Spravato by spraying it into your nose. This drug is always taken in a medical facility under the direct guidance of a doctor. They’ll provide step-by-step instructions and make sure you give yourself the dose correctly. And they’ll continue to watch you for any side effects for at least 2 hours afterward.
People who take Spravato sometimes get nauseous, so you may be advised to avoid eating for 2 hours before your dose. You may also be advised to avoid drinking any fluids for 30 minutes beforehand.
Your doctor will give you the correct number of nasal (nose) spray devices to get your full dose. For example, you’ll use three devices to get a dose of 84 milligrams (mg).
Before taking your dose, you’ll blow your nose. Then, you’ll lean your head back slightly, put the tip of the nasal spray device into one nostril, and press your other nostril closed. You’ll breathe in through your nose as you press the plunger completely up. Then you’ll breathe in again through your nose gently. The second spray will use the same device in your other nostril. You’ll then wait 5 minutes before using the next device if your dose requires it.
You can learn more about the dosing instructions for Spravato in the prescribing information.
Note: You and your doctor will need to participate in the Spravato REMS program in order to access this drug. See “What is the Spravato REMS program?” above to learn more.
The dosage of Spravato you’re prescribed may depend on several factors. These include:
- the kind and severity of the condition you’re taking Spravato to treat
- your age
- other medications you take
- your body’s response to the drug, such as side effects including:
- sedation (feeling sleepy or having difficulty thinking straight)
- dissociation (feeling disconnected from your mind or body)
- respiratory depression (breathing that is slow and ineffective)
- worsening depression
- high blood pressure
- other conditions you may have (see “Dosage adjustments” under “What is Spravato’s dosage?”)
If you have to miss a dose of Spravato, tell your doctor right away. They may adjust your dosing schedule.
Spravato is a Schedule III controlled substance with a boxed warning regarding the risk of misuse (also known as abuse). A boxed warning is the most serious warning from the Food and Drug Administration (FDA). With misuse, a drug is taken in a way other than how it’s prescribed. This can lead to dependence. (For more on dependence, see the section directly below.)
The risk of misuse with Spravato is lowered because it is given in a healthcare facility. You’ll only take Spravato under the supervision of a healthcare professional.
For more information about the potential for misuse with Spravato, see the “Boxed warning” section in the introduction of this article.
Spravato is a controlled substance. That means it’s a drug the government regulates due to the risk of misuse and dependence. With dependence, your body needs the drug to feel like it usually does. (For more information on misuse, see the section directly above.)
No withdrawal symptoms were seen up to 4 weeks after Spravato treatment was stopped in studies. Withdrawal symptoms are side effects that can occur when you stop taking a drug that your body has become dependent on.
It isn’t known whether withdrawal symptoms might occur if Spravato is stopped after being taken for a longer time or in higher dosages than what was studied.
A similar drug, ketamine, is known to cause symptoms of withdrawal, such as:
But these symptoms have not been seen in people taking esketamine (the active drug in Spravato).
Talk with your doctor about the possible risk of dependence and withdrawal with Spravato.
The sections above describe the usual dosages provided by the drug’s manufacturer. If your doctor recommends Spravato for you, they’ll prescribe the dosage that’s right for you.
Here are some examples of questions you may want to ask your doctor:
- Which activities should I avoid on the day I get my Spravato dose?
- Will my dose of Spravato be different if I also take other antidepressants?
- Will I need to reschedule my Spravato dose if I have a stuffy or runny nose that day?
To learn more about Spravato, see these articles:
To get information on different conditions and tips for improving your health, subscribe to any of Healthline’s newsletters. You may also want to check out the online communities at Bezzy. It’s a place where people with certain conditions can find support and connect with others.
Q:
Can I take Spravato at home?
AnonymousA:
No, Spravato cannot be taken at home. This drug must be taken in a medical facility that’s certified by a special safety program called the Spravato REMS program. (REMS stands for risk evaluation and mitigation strategy.)
Spravato is a nasal (nose) spray that you’ll administer to yourself under the direct supervision of your doctor (or other healthcare professional). They’ll give you step-by-step instructions and make sure you administer your dose correctly. A healthcare professional will continue to watch you for at least 2 hours each time you take Spravato to monitor for any side effects.
If you have any questions about the REMS program or how to take Spravato, talk with your doctor. You can also visit the Spravato REMS website or call 855-382-6022 for more information.
The Healthline Pharmacist TeamAnswers represent the opinions of our medical experts. All content is strictly informational and should not be considered medical advice.Disclaimer: Healthline has made every effort to make certain that all information is factually correct, comprehensive, and up to date. However, this article should not be used as a substitute for the knowledge and expertise of a licensed healthcare professional. You should always consult your doctor or another healthcare professional before taking any medication. The drug information contained herein is subject to change and is not intended to cover all possible uses, directions, precautions, warnings, drug interactions, allergic reactions, or adverse effects. The absence of warnings or other information for a given drug does not indicate that the drug or drug combination is safe, effective, or appropriate for all patients or all specific uses.